Research
Novel X-ray light sources based on plasma-wakefield acceleration
Synchrotrons and free-electron lasers (FELs) are the most powerful sources of X-ray radiation. They constitute invaluable tools for a broad range of research (medicine, biology, chemistry, physics), however, due to their kilometer-scale sizes only a few of these sources exist worldwide. In contrast, ultra-compact, laser-driven plasma-wakefield accelerators provide markedly increased accelerating fields and hence offer the potential to shrink the size and cost of these X-ray facilities to university-laboratory scale.
We have been pioneering the field of “table-top FELs” ever since laser-plasma accelerators have been emerging. In 2009 we demonstrated the world’s first laser-driven soft X-ray undulator (LUX) source based on a laser-plasma accelerator (Fuchs et al., Nature Physics, 2009).
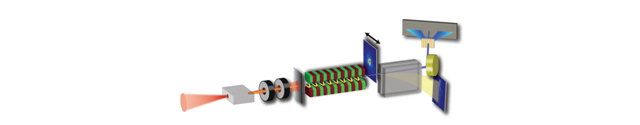
Apart from driving FEL- or LUX-sources, electrons from a laser-plasma accelerator can also drive a Thomson-source, where a part of the driver-laser pulse is scattered off the laser-accelerated electrons to emit undulator-like radiation in the hard X-ray range (50-100 keV). Currently, our team is working closely with the DESY group led by Dr. Jens Osterhoff on the establishment of such an all-optical, laser-plasma accelerator-based Thomson source for generating X-ray beams suitable for our research program on biomedical imaging. This allows for novel biomedical imaging modalities that require pencil X-ray beams, such as X-ray Fluorescence Imaging (XFI, see below). One future goal is to enable XFI in hospitals for new diagnostics for which compact sources are required.
A recent design study for such a source was published in T. Brümmer et al., Physical Review Accelerators and Beams, 2020.
Biomedical X-ray Fluorescence Imaging (XFI)
X-Ray Fluorescence Imaging (XFI) is based on the emission of characteristic X-ray fluorescence photons upon excitation of dedicated marker elements by scanning X-ray beams. This allows for tracking different biological entities, such as immune cells or medical drug compounds. To make such entities visible for XFI, they need to be labelled with XFI marker elements, such as nanoparticles or molecular tracers. For these labelling challenges, we cooperate closely with our nanoscience institute (Prof. Dr. Wolfgang Parak) and the Fraunhofer Institute CAN (Dr. Neus Feliu, Dr. Oliver Dabrowski).
A few years ago, we have presented a breakthrough by developing a computer algorithm-based method called "spatial filtering" which allows XFI even for human-sized objects. This novel approach has solved the long-standing "XFI background problem" by making use of the anisotropy of Compton-scattered photons when they exit from large objects (F. Grüner et al., Scientific Reports, 2018). This discovered anisotropy has nothing to do with the anisotropic Compton-scattering cross-section, but rather with the anisotropic yields from all paths that Compton-scattered photons can take across a given object: there are paths, and hence directions from the object relative to the incident beam, which have much lower yields of Compton-scattered photons than other paths. Figure 1 shows the comparison between simulated XFI-spectra where only the right one (b) shows detectable signals (gold fluorescence peaks in red) after the application of the spatial filtering algorithm while in the left case (a) the background level is orders of magnitude higher and hence no significant XFI signals can be measured.
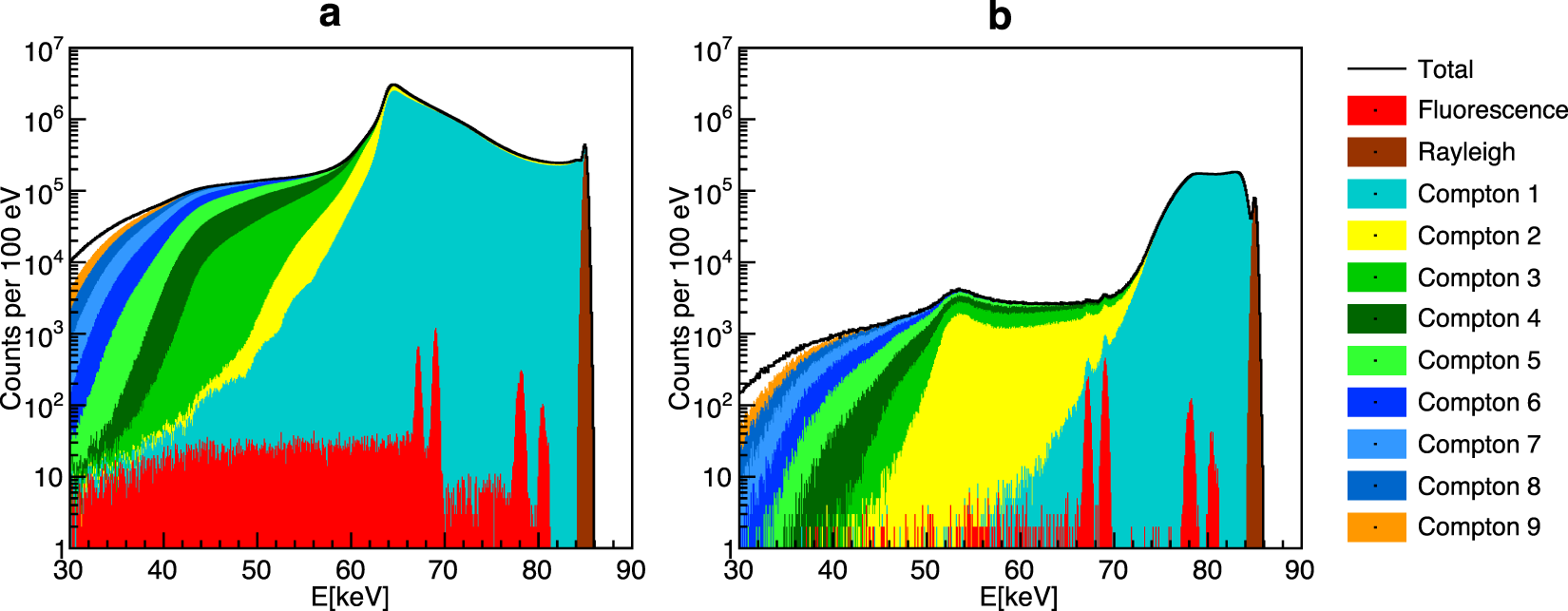
Figure 1: Simulated X-ray spectra demonstrating effective background reduction. The two fluorescence lines in red (around 69 keV) are not detectable in the left panel due to the high background level, which our filtering scheme reduces by a factor of 570, making the XFI lines visible (right panel). [Taken from our publication F. Grüner et al., Scientific Reports, 2018].
While the results presented in Figure 1 demonstrate the basic working principle of the spatial filtering algorithm when using a water sphere of 30 cm diameter with gold nanoparticles in its center, a more realistic scenario was studied using a human voxel phantom in the simulations. The findings from this study demonstrate the applicability of XFI to track immune cells labelled with gold nanoparticles in humans as exemplary shown in Figure 2 where the right and left colon are scanned.
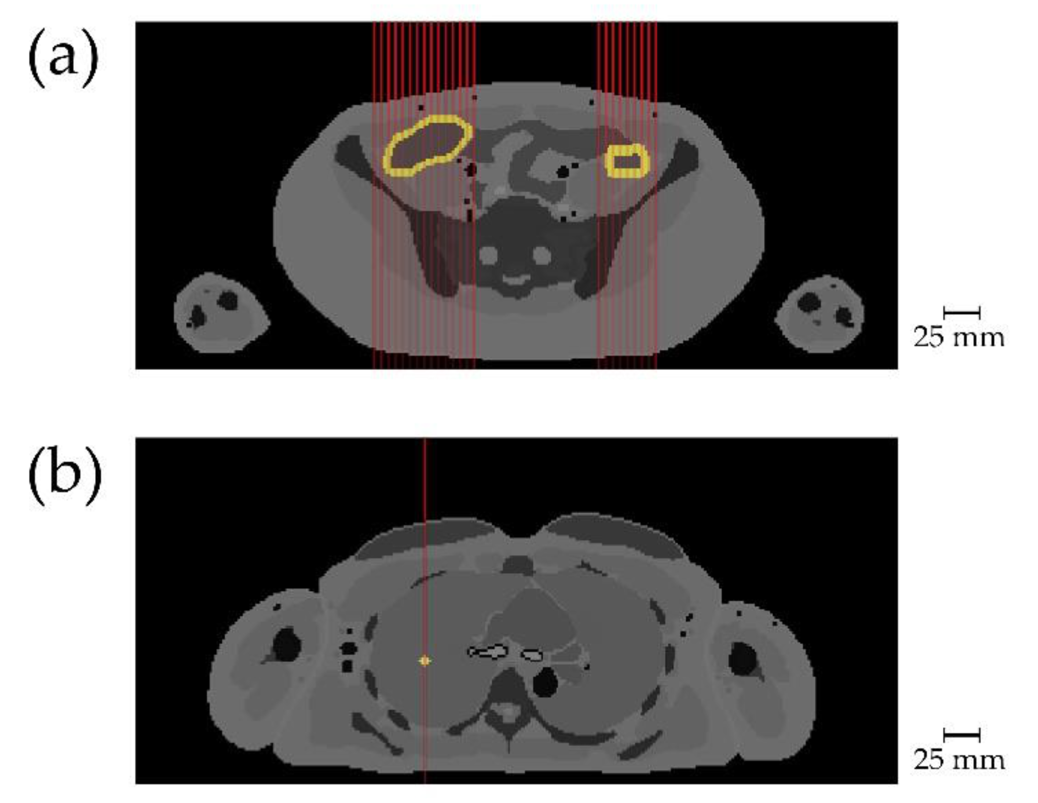
Figure 2: Target tissue with added gold is shown in light yellow, red lines indicate incident X-ray beams. [Taken from our publication A. Ungerer et al., Cancers, 2021]
The key goal of our current research, which closely involves groups from the University Hospital UKE (Prof. Dr. Samuel Huber, Prof. Dr. Udo Schumacher, Prof. Dr. Kai Rothkamm, PD Dr. Thorsten Frenzel, PD Dr. Rene Werner, and others), is to enable preclinical XFI for tracking immune cells (e.g., studying immune-mediated inflammation diseases such as Morbus Crohn) and a next-generation of pharmacokinetics (e.g., the biodistribution of cytostatics in solid tumor models). Both key applications aim at the in vivo detection of smallest local amounts of the labelled entities. The basic challenge here is to find a way to effectively reduce the intrinsic background in XFI made up by multiple Compton-scattered photons, which have in total much higher yields than the XFI signal photons. Hence, our focus is the physics of the interactions of about 50 keV photons with soft matter for finding ways of reducing their spectral contribution.
Apart from detailed computer simulations, we have performed several experimental pilot studies at DESY’s PETRA III synchrotron aiming at the detection of unprecedented small local amounts of XFI-markers (please refer to our list of publications below). In one of those measurements the before inaccessible endogenous iodine content in a murine thyroid was quantified with high resolution as shown for different mice and spatial resolutions in Figure 3.
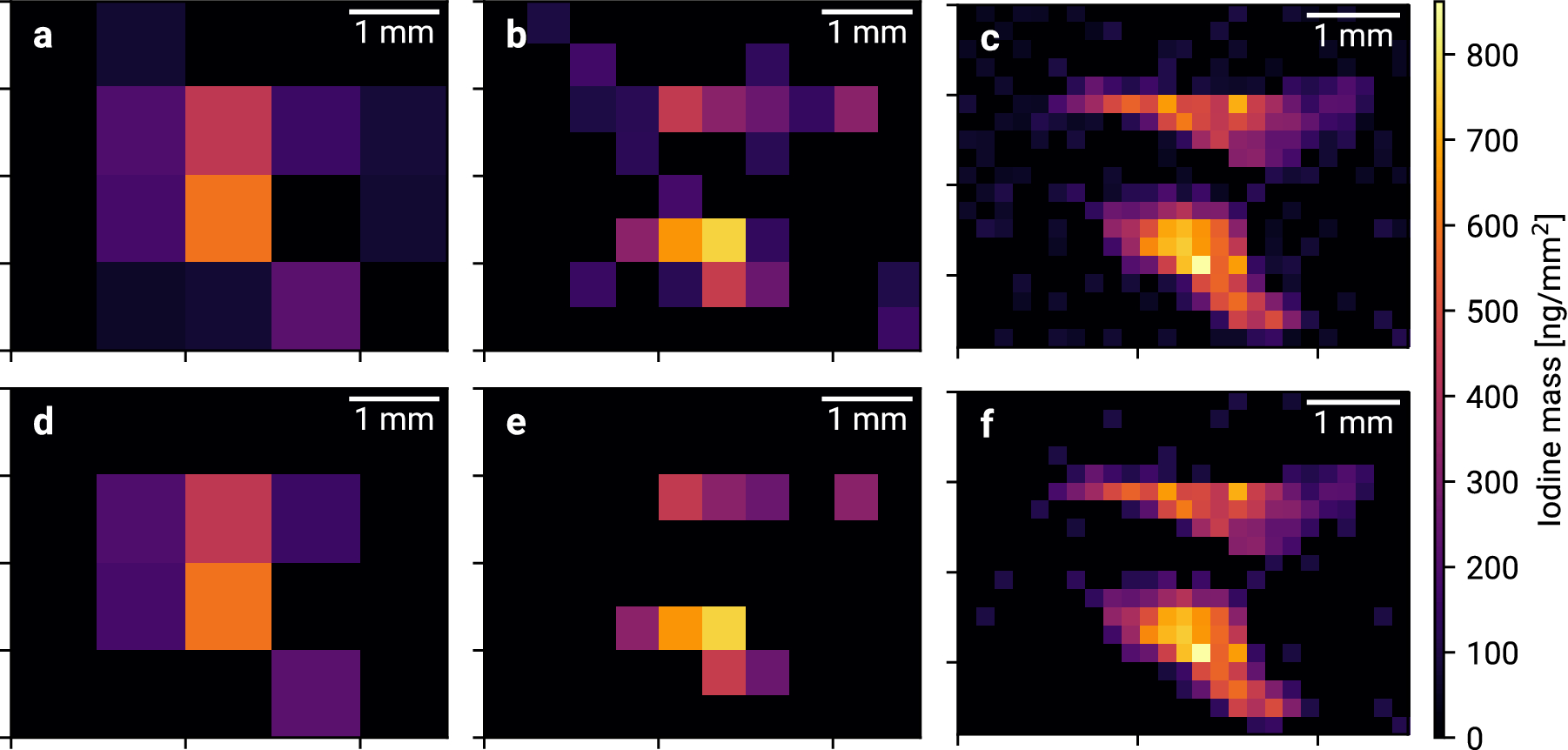
Figure 3: Thyroidal iodine mass maps for 1 mm, 0.5 mm and 0.2 mm resolution. [Taken from our publication C. Körnig et al., Scientific Reports, 2022]
One key feature of XFI is the ability to perform multi-scale imaging, meaning measurements on size scales ranging from full-body in vivo scans down to ex vivo measurements of single cells. In a cooperation with the Beiersdorf AG the uptake of iodine-labelled co-enzyme Q10 into single skin cells was studied by using tightly focused X-ray beams of the P06 beamline at PETRA III. As can be seen in Figure 4, each single cell did take up the co-enzyme and there is a clear correlation between cell size and uptake yield.

Figure 4: Images of zinc (cyan) and iodine (magenta) elemental maps for the full scan (a,b) and for the fine scan of 3 selected cells (c,d). As zinc is mainly located in the cell nucleus, its elemental map is ideally suited to indicate the locations of the nuclei. The direct comparison of iodine and zinc emphasizes the homogenous distribution of iodine among the cytoplasm. [Taken from our publication T. Staufer et al., Antioxidants, 2022].
In cooperation with the group of Prof. Hoeschen (U Magdeburg) we are currently also working on further X-ray imaging methods besides XFI which also need a pencil X-ray beam as delivered by synchrotrons and laser-based Thomson sources. In a BMBF-project together with a group from the TU Berlin the use of dedicated X-ray optics at different X-ray sources has been studied with simulations and demonstration measurements. Research on this topic will be continued to enable XFI-measurements at laboratory-based X-ray sources.
Our team provides possibilities for contract research in the area of preclinical imaging. Upon reasonable request to the group leader, we can send you a brochure with key aspects. Contract research is always negotiated and set up with the University administration, our team then plans, performs, and analyses the measurements.
Publication list:
• Florian Grüner et al. “Localising functionalised gold-nanoparticles in murine spinal cords by X-ray fluorescence imaging and background-reduction through spatial filtering for human-sized objects”, Scientific Reports, Volume 8, Issue 1, Article number 16561 (2018)
https://doi.org/10.1038/s41598-018-34925-3
• Carlos Sanchez-Cano et al. “X-ray-Based Techniques to Study the Nano–Bio Interface”, ACS Nano 2021, 15, 3754−3807
https://doi.org/10.1021/acsnano.0c09563
• Oliver Schmutzler et al. “X-ray Fluorescence Uptake Measurement of Functionalized Gold Nanoparticles in Tumor Cell Microsamples”, Int. J. Mol. Sci. 2021, 22, 3691
https://doi.org/10.3390/ijms22073691
• Henrik Kahl et al.” Feasibility of Monitoring Tumor Response by Tracking Nanoparticle-Labelled T Cells Using X-ray Fluorescence Imaging—A Numerical Study”, Int. J. Mol. Sci. 2021, 22, 8736.
https://doi.org/10.3390/ijms22168736
• A. Ungerer et al. "X-ray-Fluorescence Imaging for In Vivo Detection of Gold-Nanoparticle-Labeled Immune Cells: A GEANT4 Based Feasibility Study", Cancers 2021, 13(22):5759
https://doi.org/10.3390/cancers13225759
• C. Körnig et al. " In‐situ X‐ray fluorescence imaging of the endogenous iodine distribution in murine thyroids “, Scientific Reports 12, 2903, 2022
https://doi.org/10.1038/s41598-022-06786-4
• J. Baumann et al. “Enabling Coarse X-ray Fluorescence Imaging Scans with Enlarged Synchrotron Beam by Means of Mosaic Crystal Defocusing Optics “, Int. J. Mol. Sci. 2022, 23(9), 4673
https://doi.org/10.3390/ijms23094673
• T. Staufer, M.L. Schulze, O. Schmutzler et al. “Assessing Cellular Uptake of Exogenous Coenzyme Q10 into Human Skin Cells by X-ray Fluorescence Imaging“, Antioxidants 11, no. 8:1532, 2022
https://doi.org/10.3390/antiox11081532
• Y. Liu et al. “Size- and Ligand-Dependent Transport of Nanoparticles in Matricaria chamomilla as Demonstrated by Mass Spectroscopy and X-ray Fluorescence Imaging”, ACS Nano, 16,8, 12941–12951, 2022
https://doi.org/10.1021/acsnano.2c05339
• T. Staufer et al. “Enabling X-ray fluorescence imaging for in vivo immune cell tracking”, Scientific Reports 13, 11505, 2023
https://doi.org/10.1038/s41598-023-38536-5
• T. Staufer und F. Grüner “Review of Development and Recent Advances in Biomedical X-ray Fluorescence Imaging”, Int. J. Mol. Sci. 2023, 24(13):10990.
https://doi.org/10.3390/ijms241310990
Astrophysics in the Lab and New Paths in Gravitational Waves Detection
Recently, within the Cluster of Excellence “Quantum Universe” framework we also initiated a program on astrophysical experiments in the lab. Hence, our research group does not only set up laser-plasma accelerator experiments for driving Thomson-sources for biomedical imaging (see above), but also studies the generation of neutral electron-positron pair-beams and their beam-plasma instabilities when they move through a second plasma stage downstream, thus mimicking cosmic ray instabilities.
Our aim here is to vary relevant parameters such as the electron beam vs. ambient plasma densities in order to study the scalability of lab results onto astrophysical scales. The key challenge here is to find a way of transporting the neutral pair-beam in such a way that an inverted momentum spectrum builds up which then triggers the onset of self-amplifying beam-plasma instabilities.
There is a direct link between this laboratory astrophysics project and biomedical imaging - as for both fields advanced X-ray detectors are needed: the pair-beam generation is based upon the conversion of Bremsstrahlung-photons into electron-positron pairs, hence a precise knowledge of the Bremsstrahlung spectrum is required which can only be obtained with energy-resolved, high efficiency detectors. Such devices are also needed for the almost background-free detection of XFI-photons.
"Within "Quantum Universe" we have recently launched a second project aiming at a feasibility study on new paths in the detection of sub-Hz gravitational waves (GW), whereby the detection will be based on storage rings, possibly including laser-cooled beams. This project combines cosmology, astrophysics, and accelerator physics, and brings together different groups within our Cluster, like the University's Observatory and Accelerator Physics groups. Suvrat et al. have recently published a paper with scaling laws and first estimates on the impact of a GW on the "ticking" of particle bunches moving in a storage ring. In this feasibility study we will explore various scenarios towards identifying those which offer realistic future realizations.".
